Relationship of charge to antiaromaticity in bis-fluorenyl dications and fluorenyl monocations: experimental support for theoretical calculations†‡
Abstract
The antiaromaticity of fluorenyl
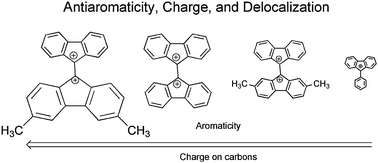
- This article is part of the themed collection: Aromaticity

 Please wait while we load your content...
Please wait while we load your content...