Mechanism of the oscillatory decomposition of the dithionite ion in a flow reactor
Abstract
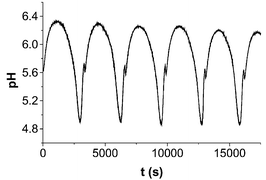
A simple mechanism consisting of three protonation equilibria and seven redox reactions between sulfur species can describe the large amplitude-sustained temporal pH-oscillations observed during acid-induced decomposition of the dithionite ion in a continuous-flow stirred tank reactor (CSTR) in the temperature range 25–60 °C.

 Please wait while we load your content...
Please wait while we load your content...