A novel competitive-displacement fluorescence assay for l-penicillamine based on the reaction between the target and N-acetyl-l-cysteine-capped CdTe quantum dots for copper ions†
Abstract
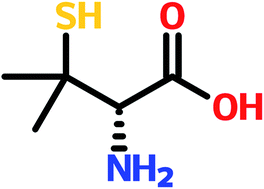
D-Penicillamine (D-PA), as the dominant enantiomer, is usually used in many clinical drugs, but the other enantiomer, L-penicillamine (L-PA) is a toxic enantiomer. Therefore, it is of great significance to analyze and detect L-PA in biomedical or environmental samples. CdTe quantum dots (CdTe QDs) were modified by N-acetyl-L-cysteine (NALC) as novel fluorescent probes and were successfully utilized for the determination of L-penicillamine. The fluorescence of the CdTe QDs decreased significantly in the presence of cupric ions (Cu2+) due to the binding of Cu2+ to NALC on the surface of the CdTe QDs, but the addition of L-PA immediately resulted in efficient fluorescence recovery of the modulated CdTe QDs. The experimental results showed that the pH of the buffer solution and the concentration of Cu2+ affected the fluorescence intensity upon adding L-PA. Under the optimum conditions, the recovered fluorescence intensity was linearly proportional to the increasing L-PA concentration in the range of 2 × 10−7 to 4 × 10−6 mol L−1, with correlation coefficient R2 = 0.9980. So, a new, simple and rapid method for the determination of L-PA has been developed and its potential has been demonstrated in human serum samples and environmental samples with satisfactory results.


 Please wait while we load your content...
Please wait while we load your content...