A sensitive and selective electrochemical nitrite sensor based on a glassy carbon electrode modified with cobalt phthalocyanine-supported Pd nanoparticles†
Abstract
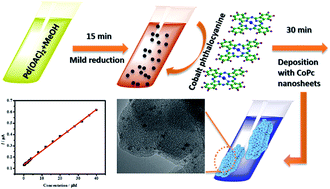
Cobalt phthalocyanine (Pd/CoPc) supported palladium nanoparticle composite materials are synthesized by using a facile methanol-mediated weakly-capping growth approach with anhydrous methanol as the mild reductant. Pd/CoPc nanorods have been used to modify a glassy carbon electrode to fabricate a sensitive and selective electrochemical nitrite sensor. The morphology and structure of Pd/CoPc are studied by XRD, UV-vis, FTIR, Raman, HRTEM and HAADF-STEM techniques. Transmission electron microscopy and high-angle annular dark-field imaging-scanning transmission electron microscopy images indicated that the diameters of spherical Pd particles are in the range of 4–6 nm. The electrode exhibits enhanced electro-catalytic behavior to the oxidation of nitrite compared to commercial activated carbon-supported palladium and pure cobalt phthalocyanine (CoPc) modified glassy carbon electrodes. Meanwhile, due to the unique structure of the Pd/CoPc modified electrode, the prepared nitrite biosensor displays good performance. The fabricated sensor exhibits excellent analytical performance for nitrite detection linearly over two different concentration ranges, which are from 0.2 to 50 μM and from 500 to 5000 μM with a low detection limit of 0.10 μM nitrite and a sensitivity of 0.01 μA μM−1 (S/N = 3). Moreover, the proposed electrochemical sensor also shows good reproducibility and excellent anti-interference ability against metal ions, and exhibits satisfactory recovery for detecting nitrite in cured food.


 Please wait while we load your content...
Please wait while we load your content...