Electrochemical chip integrating scalable ring–ring electrode array to detect secreted alkaline phosphatase†
Abstract
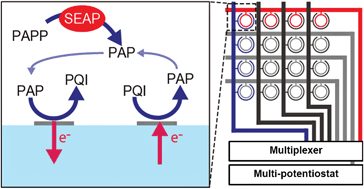
An electrochemical platform for parallel monitoring of secreted alkaline phosphatase (SEAP) has been microfabricated on a device with a mammalian-cell array chip. A 4 × 4 ring–ring

 Please wait while we load your content...
Please wait while we load your content...