Organocatalytic atroposelective synthesis of naphthoquinone thioglycosides from aryl-naphthoquinones and thiosugars†
Abstract
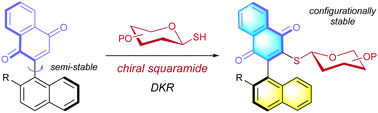
In this study, we report an organocatalytic formal coupling strategy for aryl-naphthoquinones with thiosugars that provides straightforward access to the axially chiral naphthoquinone thioglycoside with excellent stereoselectivity. Mechanistic studies revealed the key role of H-bonding in stereochemical recognition. The reaction pathway involves the atroposelective addition, followed by stereoretentive oxidation of the hydroquinone intermediate.


 Please wait while we load your content...
Please wait while we load your content...