Chiral phosphoric acid-catalyzed regio- and enantioselective reactions of functionalized propargylic alcohols†
Abstract
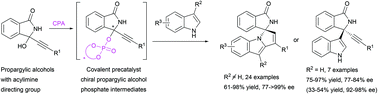
Propargylic alcohols have been known as useful substrates in a wide range of asymmetric reactions. In particular, chiral phosphoric acid (CPA) catalyzed reactions of functionalized propargylic alcohols have opened robust access to axially chiral tetrasubstituted allenes and their related derivatives. However, these types of propargylic alcohols have been essentially limited to the structures that could form quinone methides or methide indoles. Here, we expand the scope of propargylic alcohols and the power of CPA catalysis. With the established system, racemic 3-alkynyl-3-hydroxyisoindolinones reacted efficiently to form diverse spirocyclic heterocycles with high enantioselectivity. Regiodivergence was also observed with different indole nucleophiles. Importantly, the key covalently bonded CPA adduct was isolated, characterized, and further confirmed to be chemically competent, and a possible covalent activation mode was proposed accordingly.
- This article is part of the themed collection: 2022 Organic Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...