An investigation of palladium-catalyzed Stille-type cross-coupling of nitroarenes in perylenediimide series†
Abstract
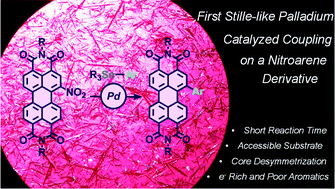
We report herein an unprecedented palladium-catalyzed cross-coupling reaction between mononitro-perylenediimide (PDI) and various arylstannanes. Optimized conditions developed with this Stille-type reaction allow the grafting of (hetero)aryls of various electronic nature in the bay region of PDIs. Moreover, we capitalized on the high selectivity of this cross-coupling through the desymmetrization of the dinitro-PDI substrate.


 Please wait while we load your content...
Please wait while we load your content...