A new diformyl phenol based chemosensor selectively detects Zn2+ and Co2+ in the nanomolar range in 100% aqueous medium and HCT live cells†
Abstract
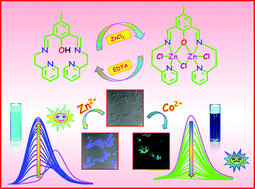
In this manuscript, we present a chemosensor, 4-methyl-2,6-bis-[(2-pyridin-2-yl-ethylimino)-methyl]-phenol (Hmpye), for selective detection of Zn2+ and Co2+ in 100% aqueous medium. The active component of the sensor is the Schiff base condensation product between 4-methyl-2,6-diformylphenol and 2,2-pyridyl ethylamine and the resultant moiety is ESIPT (excited-state intramolecular proton transfer) active via imine nitrogen and phenolic OH. The absorption and emission spectral investigations have been carried out extensively for the sensing system which showed that Hmpye is able to sense Zn2+ by fluorescence turn-on and Co2+ by fluorescence turn-off. Apart from Job's plot and ESI-MS studies, the single crystal X-ray diffraction study has been utilized to ascertain the 1 : 2 ratio of the sensor : Zn2+ ensemble. The detection limits for Zn2+ and Co2+ are 4.683 × 10−9 (M) and 7.591 × 10−9 (M), respectively. The sensing capability of Hmpye has also been checked in HCT live cells. The potential of the work has been demonstrated by the fact that nanomolar detection of Zn2+ and Co2+ from 100% aqueous solution can be achieved by a single probe which is scarce in the literature.


 Please wait while we load your content...
Please wait while we load your content...