Side-chain alkylation of toluene with methanol over cesium-ion-exchanged zeolite LSX and X catalysts†
Abstract
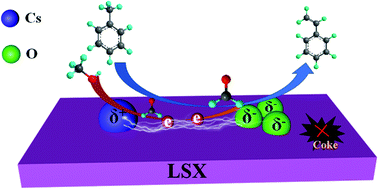
A low-silica X (Si/Al = 1.01, LSX) zeolite was successfully synthesized through a two-step hydrothermal method. It was then modified with Cs+ using the ion-exchange method, and the obtained sample was tested as a catalyst in the side-chain alkylation of toluene with methanol. Compared with conventional CsX catalyst, the CsLSX catalyst exhibited improved reactivity in terms of toluene conversion. The toluene conversion was 8.50% on the CsLSX catalyst. One reason for this is that the CsLSX with a low Si/Al ratio had a stronger capacity for basic cation exchange. The greater amount of alkali cations promoted the dehydrogenation of methanol to formaldehyde, which was beneficial for the side-chain alkylation of toluene. Additionally, the higher number of aluminum atoms on the CsLSX catalyst allowed it to have more oxygen atoms with a negative charge (Oδ−) in the zeolite framework. These were more favorable for adsorbing toluene molecules and more effectively activated the C–H bond in the methyl side-chain of toluene. In addition, the CsLSX catalyst also exhibited improved catalytic stability, and enhanced selectivity toward styrene was obtained over CsLSX further modified with Na2B4O7 or ZrB2O5.


 Please wait while we load your content...
Please wait while we load your content...