Synthesis of oxazolidinones through ring-opening and annulation of vinylene carbonate with 2-pyrrolyl/indolylanilines under Rh(iii) catalysis†
Abstract
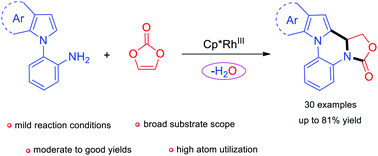
Herein, we have developed a rhodium-catalyzed C–H functionalization and subsequent intramolecular ring-opening/cyclization of vinylene carbonate with 2-pyrrolyl/indolylanilines, which leads to oxazolidinones in moderate to good yields. In this transformation, vinylene carbonate only eliminates one oxygen atom rather than –CO3 or CO2. Furthermore, some control experiments are conducted to elucidate the reaction mechanism.


 Please wait while we load your content...
Please wait while we load your content...