Synthesis and characterization of CO2-sensitive temperature-responsive catalytic poly(ionic liquid) microgels†
Abstract
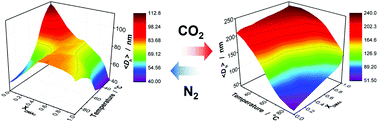
We report a class of poly(anionic liquid) microgels that are sensitive to carbon dioxide (CO2). Such microgels have been synthesized from free radical precipitation polymerization of tetrabutylphosphonium 4-styrenesulfonate, 1-vinyl-1H-imidazole and the crosslinker N,N′-methylenebisacrylamide. Upon bubbling with N2, these microgels can undergo reversible volume phase transition in response to a change in temperature, showing a lower critical solution temperature (LCST) in water, but showing an upper critical solution temperature (UCST) in dimethylacetamide, such that the temperature-responsive volume phase transition behavior can be readily tuned over a significant wide range in a mixture of water/dimethylacetamide by varying the mol ratio of the two solvents, resulting in a phase diagram displaying a co-solvency phase separation phenomenon; however, upon bubbling with CO2, they obey LCST-type volume phase transition behavior in both water, dimethylacetamide, and water/dimethylacetamide mixtures. Moreover, these microgels behave as catalysts in a model cycloaddition of CO2 to 1,2-epoxybutane in dimethylacetamide under atmospheric pressure (1 atm CO2) and display merits of both homogeneous (considerable high efficient catalytic activity) and heterogeneous (excellent recyclability) catalysis. With both the responsive and catalytic properties harnessed on the same object, the microgels react to external stimuli and allow catalytic properties to be altered accordingly to a certain extent in a non-monotonous way, making it possible to boost the reaction at a relatively lower temperature (e.g., ca. 50 °C) while maintaining considerable catalytic activity.


 Please wait while we load your content...
Please wait while we load your content...