Chemical synthesis of culmorin metabolites and their biologic role in culmorin and acetyl-culmorin treated wheat cells†
Abstract
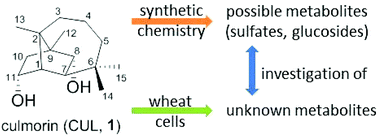
The Fusarium metabolite culmorin (1) is receiving increased attention as an “emerging mycotoxin”. It co-occurs with trichothecene mycotoxins and potentially influences their toxicity. Its ecological role and fate in plants is unknown. We synthesized sulfated and glucosylated culmorin conjugates as potential metabolites, which are expected to be formed in planta, and used them as reference compounds. An efficient procedure for the synthesis of culmorin sulfates was developed. Diastereo- and regioselective glucosylation of culmorin (1) was achieved by exploiting or preventing unexpected acyl transfer when using different glucosyl donors. The treatment of a wheat suspension culture with culmorin (1) revealed an in planta conversion of culmorin into culmorin-8-glucoside (6) and culmorin acetate, but no sulfates or culmorin-11-glucoside (7) was found. The treatment of wheat cells with the fungal metabolite 11-acetylculmorin (2) revealed its rapid deacetylation, but also showed the formation of 11-acetylculmorin-8-glucoside (8). These results show that plants are capable of extensively metabolizing culmorin.


 Please wait while we load your content...
Please wait while we load your content...