Dual responsive salen-type Schiff bases for the effective detection of l-arginine via a static quenching mechanism†
Abstract
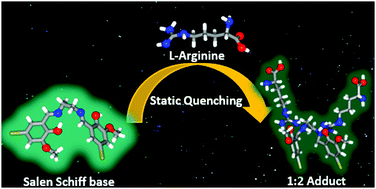
L-Arginine is one of the most important amino acids and plays a key role in diagnostic studies. Herein, we report four salen-type Schiff base fluorimetric/colorimetric sensors for the rapid detection of L-arginine. Substituents play a major role in the sensing mechanism of probes with the nitro-substituent bearing probe showing the highest efficiency. A 1 : 2 complexation between all the probes and the analyte was evidenced from the absorption titrations; and the fluorescence studies revealed a quenching phenomenon occurring during complex formation. From the life time measurements, the nature of quenching was found to be static and the formation of a ground state complex was confirmed. DFT calculations done at the B3LYP/TZVP level of theory corroborated the experimental findings and further established the proton transfer from the probe to the analyte during the complex formation.


 Please wait while we load your content...
Please wait while we load your content...