Free energy calculations and molecular properties of substrate translocation through OccAB porins†
Abstract
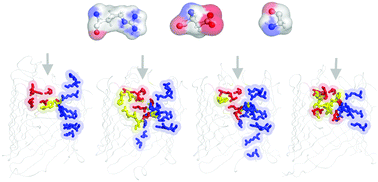
One of the greatest health threats facing modern medicine is the emergence of new bacterial strains which are increasingly resistant to almost all currently available antibiotics. According to a CDC (Center for Disease Control and Prevention) report published in 2013, 63% of Acinetobacter species have been identified as Multidrug resistant strains. As for other Gram-negative bacteria, the presence of an outer membrane increases the intrinsic resistance of A. baumannii to most antibiotics. The outer membrane of A. baumannii possesses several specific porins that control the selectivity for different polar substrates in a way that is still poorly understood. Recently, the X-ray crystal structures of 4 related porins, termed OccAB1–4, were solved at high resolution, providing a framework to study the structural and functional characteristics of these porins in filtering natural substrates. Here, we first use molecular dynamics simulations on OccAB proteins to investigate the stability and dynamics of the pores, and to establish their common biophysical features. We then applied metadynamics simulations to evaluate the free energy costs required for polar substrates to overcome the pore. Together, the comparative analysis of the OccAB porins not only sheds light on how these channels could function as potential antibiotic gateways, but also allows identification of putative affinity sites that represent a common path through which other molecules can transit.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...