Synergistic effect of the bifunctional polydopamine–Mn3O4 composite electrocatalyst for vanadium redox flow batteries†
Abstract
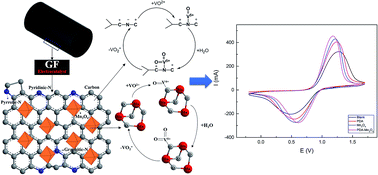
In this work, a novel catalyst based on the polydopamine (PDA)–Mn3O4 composite was synthesized and optimized by varying PDA and Mn3O4 loadings. PDA was used to immobilize Mn3O4 nanoparticles due to its abundant functional groups and self-polymerization properties. Bifunctional PDA acts as not only a bridge between Mn3O4 nanoparticles and graphite felt (GF) but also a catalyst for the VO2+/VO2+ redox reaction. The composite electrocatalyst significantly improves the electrochemical performance of GF on which the VO2+/VO2+ redox reaction tends to be sluggish. Interestingly, it was found that there was a synergistic effect in the PDA–Mn3O4 composite which resulted in better electrochemical catalytic activity than pure PDA or pure Mn3O4. The electrochemical kinetics of the optimum PDA–Mn3O4 GF with 4 h PDA polymerization time and 50 ppm Mn3O4 was investigated by cyclic voltammetry and the results showed that the diffusion coefficient of PDA–Mn3O4 GF was approximately twice that of blank GF. As expected, the optimum composite GF exhibited substantially improved performance in the vanadium redox flow battery (VRFB) single cell test. The electrolyte utilization of the VRFB with PDA–Mn3O4 GF was 94% while that of the VRFB with blank GF was 85%. The voltage efficiency (VE) and energy efficiency (EE) of the VRFB with PDA–Mn3O4 GF were increased by 5.9% and 5.2%, respectively at high current density (150 mA cm−2) compared to those of blank GF due to the synergistic effect of PDA–Mn3O4 GF. Moreover, PDA–Mn3O4 GF showed outstanding stability in both the stability test and VRFB single cell operation due to the strong interaction between PDA and Mn3O4. Compared with previously reported electrode materials based on modified GF, the novel catalyst described in this work exhibited higher energy efficiency at comparable or higher current density. Furthermore, the superior stability of Mn3O4 on the electrode with the use of bifunctional PDA could improve the lifetime of VRFBs by hindering catalyst loss and dissolution during cycles.


 Please wait while we load your content...
Please wait while we load your content...