In situ Rb–Sr and K–Ca dating by LA-ICP-MS/MS: an evaluation of N2O and SF6 as reaction gases
Abstract
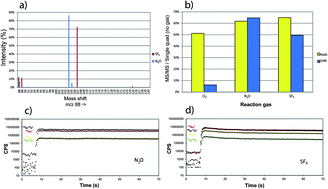
In situ dating of K-rich minerals, e.g. micas and K-feldspar, by the Rb–Sr isotopic system is a new development made possible by the ICP-MS/MS technique. Online chemical separation of Rb and Sr is possible in an O2-filled reaction cell, wherein a portion of the Sr reacts to SrO+ while simultaneously no RbO+ is formed. O2 reactions provide stable analytical conditions sufficient for precise and accurate determination of Rb/Sr and Sr/Sr isotopic ratios using 80 micron laser ablation spots. However, to date <10% of the Sr reacts with O2 as reaction gas, leaving room for improvement using more potent reaction gases. With a more efficient reactive transfer, it should be possible to obtain similar results with a smaller laser spot size, hence gaining higher spatial resolution. In this study, we have evaluated N2O and SF6 as reaction gases since they have previously been shown to react strongly with Sr+, without affecting Rb+. Analytical conditions, including cell parameters and reaction gas flow rate were optimized while ablating NIST SRM 610. The main reaction product is SrO+ for N2O reaction and SrF+ for SF6 reaction. Both gases show significantly higher reaction product formation compared to O2 with >85% of Sr reacting with N2O and >70% Sr reacting with SF6; Rb does not react with either gas. As a result, the sensitivity for Sr reaction products is ∼10 times higher with N2O and ∼8 times higher with SF6 compared to O2. With these more reactive gases, the error of mica isochron ages, calibrated against a newly developed nano-particulate pressed powder tablet of mica–Mg, is ∼1% using a 50 μm laser spot. Our tests show that both N2O and SF6 form interfering reaction products, e.g., SrOH (N2O), SiF3 and TiF3 (SF6) that are difficult to handle using single mass spectrometer instruments, but which can be overcome using MS/MS. Using SF6 combined with H2, it is possible to measure 40Ca+ as 40Ca19F+, free from interference of 40Ar+ and 40K+. This facilitates the dating of micas by the K–Ca isotopic system; we present the first in situ K–Ca age determination.
- This article is part of the themed collection: European Workshop on Laser Ablation


 Please wait while we load your content...
Please wait while we load your content...