π-Expanded 1,3-diketones – synthesis, optical properties and application in two-photon polymerization†
Abstract
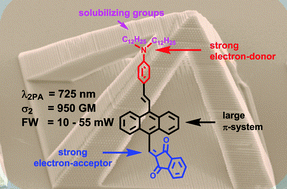
Four π-expanded α,β-unsaturated 1,3-diketones have been prepared via attaching strong electron-donating and electron-withdrawing groups at positions 9 and 10 of the anthracene scaffold. The strategic incorporation of (C12H25)2N groups at the periphery of these D–π–A molecules resulted in dyes with excellent solubility in most organic solvents. These non-planar diketones possess very broad absorption of light and negligible fluorescence. The two-photon absorption cross-section was measured via Z-scan as well as by two-photon excited fluorescence. The results clearly confirm that, in the case of compounds possessing such small fluorescence quantum yields, the Z-scan was a more reliable approach. Depending on the chemical structure, these compounds exhibited two-photon absorption cross-sections (σ2) up to 2500 GM at 725 nm. For the first time an α,β-unsaturated ketone derived from a proton sponge has been synthesized and was shown to possess optical features distinct from its simpler analogs, such as weak emission. All studied ketones possessed two-photon absorption cross-sections ∼200 GM at wavelength of two-photon polymerization (i.e. 800 nm) and broad fabrication windows have been achieved using all these dyes as photoinitiators.


 Please wait while we load your content...
Please wait while we load your content...