A genomics-led approach to deciphering the mechanism of thiotetronate antibiotic biosynthesis†
Abstract
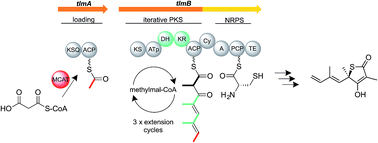
Thiolactomycin (TLM) is a thiotetronate antibiotic that selectively targets bacterial fatty acid biosynthesis through inhibition of the β-ketoacyl-acyl carrier protein synthases (KASI/II) that catalyse chain elongation on the type II (dissociated) fatty acid synthase. It has proved effective in in vivo infection models of Mycobacterium tuberculosis and continues to attract interest as a template for drug discovery. We have used a comparative genomics approach to uncover the (hitherto elusive) biosynthetic pathway to TLM and related thiotetronates. Analysis of the whole-genome sequence of Streptomyces olivaceus Tü 3010 producing the more ramified thiotetronate Tü 3010 provided initial evidence that such thiotetronates are assembled by a novel iterative polyketide synthase-nonribosomal peptide synthetase, and revealed the identity of other pathway enzymes, encoded by adjacent genes. Subsequent genome sequencing of three other thiotetronate-producing actinomycetes, including the Lentzea sp. ATCC 31319 that produces TLM, confirmed that near-identical clusters were also present in these genomes. In-frame gene deletion within the cluster for Tü 3010 from Streptomyces thiolactonus NRRL 15439, or within the TLM cluster, led to loss of production of the respective thiotetronate, confirming their identity. Each cluster houses at least one gene encoding a KASI/II enzyme, suggesting plausible mechanisms for self-resistance. A separate genetic locus encodes a cysteine desulfurase and a (thiouridylase-like) sulfur transferase to supply the sulfur atom for thiotetronate ring formation. Transfer of the main Tü 3010 gene cluster (stu gene cluster) into Streptomyces avermitilis led to heterologous production of this thiotetronate, showing that an equivalent sulfur donor can be supplied by this host strain. Mutational analysis of the Tü 3010 and TLM clusters has revealed the unexpected role of a cytochrome P450 enzyme in thiotetronate ring formation. These insights have allowed us to propose a mechanism for sulfur insertion, and have opened the way to engineering of the biosynthesis of TLM and other thiotetronates to produce novel analogues.

 Please wait while we load your content...
Please wait while we load your content...