Photo-responsive modulation of hybrid peptide assembly, charge transfer complex formation and gelation†
Abstract
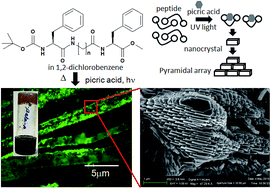
The photo-responsive modulation of self-assembly and charge transfer complex formation of a series of hybrid peptides Boc–Phe–Xaa–Phe–OMe (Xaa = glycine/β-alanine/γ-aminobutyric acid) with differently spaced diphenylalanine have been investigated. In presence of picric acid the tripeptides containing β-alanine and γ-aminobutyric acid form photo responsive charge-transfer complex and organogels. 366 nm UV irradiation acts as a source of energy for the modulation and rearrangement of π-stacking in the supramolecular systems. The fluorescence spectroscopy shows a red shift for this photo-induced charge-transfer complex formation. The photo-induced structural changes have been studied by CD and FT-IR spectroscopy. Fluorescence microscopy and field emission scanning electron microscopy of the xerogels exhibit formation of micro crystals array by photo induced charge-transfer complex formation. But the peptide 1 containing a core of conformationally flexible achiral glycine and two termini of phenylalanine residues does not form photo responsive organogel. The X-ray crystallography reveals that the peptide 1 adopts parallel β-sheet conformation which further self-associates to form parallel sheet-like structure through multiple intermolecular hydrogen bonding and π-stacking interactions. The achiral homologues spacers between two phenylalanine residues play a key role for the photo-responsive assembly process.

 Please wait while we load your content...
Please wait while we load your content...