Synthesis and electrochemical performance of Co2TiO4 and its core–shell structure of Co2TiO4@C as negative electrodes for Li-ion batteries
Abstract
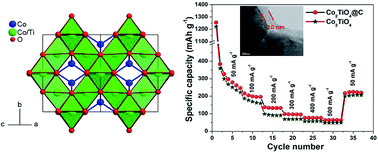
Spinel Co2TiO4 is synthesised using a polymeric precursor method and used as an efficient negative electrode for Li-ion batteries. Precise full-profile Rietveld refinement proves the formation of a single-phase cubic spinel structure with a lattice parameter of a = 8.4190(9) Å, which corresponds to the sample composition of Co2.05Ti0.95O4. Subsequently, a carbon coating around Co2TiO4 is achieved through a simple hydrothermal method. TGA analysis implies that Co2TiO4@C consists of 17 wt% carbon, and the presence of D and G bands was confirmed through Raman analysis. Transmission electron microscopy (TEM) is employed to probe the morphological features, as well as to confirm the carbon coating on Co2TiO4. It shows non uniform shape particles with sizes in the range of 400–750 nm and that the thickness of the carbon coating is 10 nm. The superior electrochemical performance of Co2TiO4@C is confirmed by a higher initial discharge–charge capacity (1283/418 mA h g−1), high diffusion coefficient (1.76 × 10−10 cm2 s−1/2) and lower Rct (after 50 cycles). This is attributed to the increased electrical conductivity and the creation of new active sites due to the synergistic effect of the carbon matrix on Co2TiO4, thereby making it a promising candidate for lithium ion battery applications.

 Please wait while we load your content...
Please wait while we load your content...