Enantioselective syntheses of β-amino alcohols catalyzed by recyclable chiral Fe(iii) metal complex†
Abstract
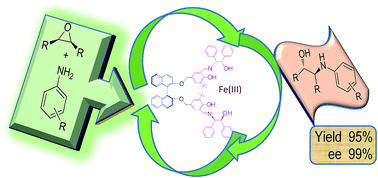
An efficient asymmetric desymmetrization of meso-epoxides with anilines catalysed by a series of simple and environmentally benign in situ generated Fe(III) complexes based on chiral tridentate ligands L1–L7 with achiral and chiral linkers (methylene, piperazine, R/S BINOL and diethyl tartrate) was carried out at rt. The in situ generated iron metal complex based on ligand L5a emerged as improved (low catalyst loading) catalyst for asymmetric desymmetrization of meso-epoxides with anilines giving high enantioselectivity (up to 99%) and high yield (95%) of enantiopure β-amino alcohols in 14 h. While excellent results for ARO of cyclic as well as aliphatic epoxides with anilines was achieved with in situ generated complex from the ligand L4h and Fe(III) chloride, the catalyst was recoverable and recyclable (five times) with retention of its performance.

 Please wait while we load your content...
Please wait while we load your content...