Monodispersed ultramicroporous semi-cycloaliphatic polyimides for the highly efficient adsorption of CO2, H2 and organic vapors†
Abstract
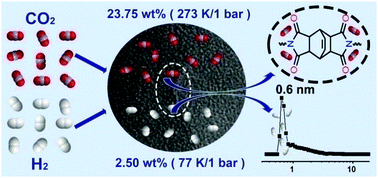
Ultramicroporous semi-cycloaliphatic polyimides (sPIs) have been synthesized via solution polycondensation from bicyclo[2.2.2]oct-7-ene-2,3,5,6-tetracarboxylic dianhydride with 1,3,5,7-tetrakis(4-aminophenyl)adamantane and tetrakis(4-aminophenyl)methane, respectively. The well-controlled kinetic conditions result in a monodispersed pore size of 0.6 nm, located in the ultra-microporous region. Moreover, the introduction of cycloaliphatic moieties decreases the concentration of benzene rings, disrupts the π electron delocalization across the conjugated polyimide backbone and thereby effectively reduces the charge-transfer (CT) interactions between the neighboring polyimide segments. Thus, the improved porous environment gives rise to remarkably large adsorption capacities for both gases and organic vapors. For example, sPIs can uptake 23.75 wt% CO2 (273 K, 1 bar), 2.50 wt% H2 (77 K, 1 bar), 176.0 wt% benzene and 85.1 wt% cyclohexane (298 K, P/P0 = 0.9), which are among the highest values for microporous organic polymers reported to date. In addition, it is interesting to observe that the adsorbed ratio of benzene to cyclohexane in the sPIs was tunable using various concentrations of cycloaliphatic and phenyl groups in the polyimide networks.

 Please wait while we load your content...
Please wait while we load your content...