Arylamino radical complexes of ruthenium and osmium: dual radical counter in a molecule†
Abstract
Radical and non-radical ruthenium and osmium complexes of 1-amino-9,10-anthraquinone (AqNH2), which is defined as a molecule of dual radical counter, are disclosed. 1-Amido-9,10-anthraquinone (AqNH−) complexes of the types trans-[RuII(AqNH−)(PPh3)2(CO)Cl] (1), trans-[OsII(AqNH−)(PPh3)2(CO)Br] (2) and trans-[RuIII(AqNH−)(PPh3)2Cl2] (3) were isolated. AqNH− of 1–3 is redox active and undergoes oxidation reversibly at +(0.05–0.35) V to the 1-amino-9,10-anthraquinone radical (AqNH˙) and reduction at -(0.86–1.60) V to the 1-amido-9,10-anthrasemiquinonate anion radical (AqNHSQ˙2−). The reaction of 2 with I2 in CH2Cl2 afforded a crystalline AqNH˙ complex of the type trans-[OsII(AqNH˙)(PPh3)2(CO)Br]+I5−·½I2 (2+I5−·½I2). AqNH˙ and AqNHSQ˙2− complexes of the types trans-[RuII(AqNH˙)(PPh3)2(CO)Cl]+ (1+), trans-[RuIII(AqNH˙)(PPh3)2Cl2]+ (3+), trans-[RuII(AqNHSQ˙2−)(PPh3)2(CO)Cl]− (1−) and trans-[OsII(AqNHSQ˙2−)(PPh3)2(CO)Br]− (2−) were generated chemically/electrochemically in solution. The electronic states of the complexes were authenticated by single crystal X-ray structure determinations of 1, 2·5/4 toluene, 3 and 2+I5−·½I2, EPR spectroscopy and density functional theory (DFT) calculations. AqNH˙ instigates a 2c–3e pπ–dπ interaction and the 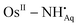 length in 2+I5−·½I2, 1.978(5) Å, is relatively shorter than the OsII–NHAq− length, 2.037(2) Å, while the Aq–NH˙ bond, 1.365(8) Å, is longer than the Aq–NH− bond, 1.328(3) Å. DFT calculations predicted that the atomic spin is delocalized over the ligand backbone (1+, 56%) particularly in one of the p-orbitals of the nitrogen and the metal atoms of the 1+ and 2+ ions, while the spin is dominantly localized on the anthraquinone fragment of the 1− and 2− ions. TD DFT calculations were employed to elucidate the origins of the lower energy absorption bands of the neutral complexes. Hypsochromic shifts of the UV-vis-NIR absorption maximum during 1→1+, 2→2+ and 3→3+ conversions were recorded by spectroelectrochemical measurements.
length in 2+I5−·½I2, 1.978(5) Å, is relatively shorter than the OsII–NHAq− length, 2.037(2) Å, while the Aq–NH˙ bond, 1.365(8) Å, is longer than the Aq–NH− bond, 1.328(3) Å. DFT calculations predicted that the atomic spin is delocalized over the ligand backbone (1+, 56%) particularly in one of the p-orbitals of the nitrogen and the metal atoms of the 1+ and 2+ ions, while the spin is dominantly localized on the anthraquinone fragment of the 1− and 2− ions. TD DFT calculations were employed to elucidate the origins of the lower energy absorption bands of the neutral complexes. Hypsochromic shifts of the UV-vis-NIR absorption maximum during 1→1+, 2→2+ and 3→3+ conversions were recorded by spectroelectrochemical measurements.


 Please wait while we load your content...
Please wait while we load your content...