Reductive nitrosylation of nickel(ii) complex by nitric oxide followed by nitrous oxide release†
Abstract
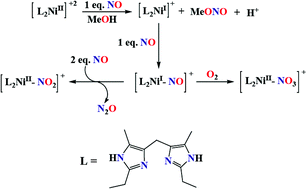
Ni(II) complex of ligand L (L = bis(2-ethyl-4-methylimidazol-5-yl)methane) in methanol solution reacts with an equivalent amount of NO resulting in a corresponding Ni(I) complex. Adding further NO equivalent affords a Ni(I)-nitrosyl intermediate with the {NiNO}10 configuration. This nitrosyl intermediate upon subsequent reaction with additional NO results in the release of N2O and formation of a Ni(II)-nitrito complex. Crystallographic characterization of the nitrito complex revealed a symmetric η2-O,O-nitrito bonding to the metal ion. This study demonstrates the reductive nitrosylation of a Ni(II) center followed by N2O release in the presence of excess NO.

 Please wait while we load your content...
Please wait while we load your content...