Mapping reactivities of aromatic models with a lignin disassembly catalyst. Steps toward controlling product selectivity†
Abstract
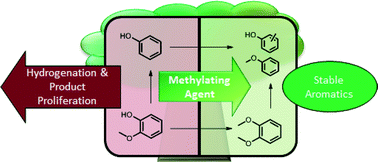
Copper-doped porous metal oxides catalyze the one-pot disassembly of biomass-derived lignin via C–O bond hydrogenolysis and hydrodeoxygenation in supercritical methanol. This catalytic system cleanly converts lignin as well as lignocellulose composites, such as sawdust, to organic liquids with little or no formation of intractable tars or chars. However, this catalyst based on Earth-abundant components also catalyzes less desirable aromatic ring hydrogenations and various methylations that contribute to the diversity of products. In this context, we undertook a quantitative experimental and computational evaluation of model reactions relevant to the reductive disassembly of lignin by this catalyst system in order to determine quantitatively the rates of desirable and less desirable chemical steps that define the overall product selectivities. Global fitting analysis methods were used to map the temporal evolution of key intermediates and products and to elucidate networks that provide guidelines regarding the eventual fates of reactive intermediates in this catalysis system. Phenolic compounds display multiple reaction pathways, but substrates such as benzene, toluene, and alkyl- and alkoxy-substituted aromatics are considerably more stable under these conditions. These results indicate that modifying this catalytic system in a way that controls and channels the reactivity of phenolic intermediates should improve selectivity toward producing valuable aromatic chemicals from biomass-derived lignin. To this end we demonstrate that the O-methylating agent dimethyl carbonate can intercept the phenol intermediate formed from hydrogenolysis of the model compound benzyl phenyl ether. Trapping the phenol as anisole thus gave much higher selectivity towards aromatic products.

 Please wait while we load your content...
Please wait while we load your content...