The oxygen reduction reaction (ORR) on reduced metals: evidence for a unique relationship between the coverage of adsorbed oxygen species and adsorption energy†
Abstract
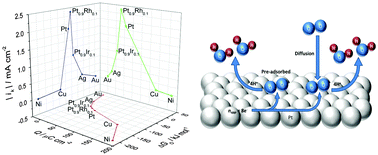
Using chronoamperometry and voltammetry at oxide-free microelectrodes in non-adsorbing electrolyte, KClO4, we investigated the reduction of oxygen containing species which adsorb at potentials below those where oxides are grown electrochemically when the electrode is exposed to dissolved oxygen. Their coverage is found to vary with the metal substrate as follows, Ni > Cu > Pt > Ag, and is negligible on Au microdiscs. This dependence is consistent with the adsorption energy of atomic oxygen for the respective metals predicted by DFT. Furthermore the adsorption energies derived from the stripping peak potentials for the different metals agree very well with those predicted by the DFT calculations. We exploit the oxygen coverage differences between Pt, Pt0.9Rh0.1 and Pt0.9Ir0.1 microdiscs to predict the adsorption energy of these species on the Pt alloys and demonstrate a clear correlation between the metal activity towards the ORR and the charge associated with the reduction of the adsorbed oxygen species. In effect the coverage – adsorption energy relationship turns the charge associated with the reduction of the adsorbed oxygen species into a descriptor of the electrode activity towards the ORR. This study provides new insight into the oxygen reduction reaction and offers a new methodology to investigate low temperature fuel cell ORR catalysts.

 Please wait while we load your content...
Please wait while we load your content...