Charging and aggregation of latex particles in aqueous solutions of ionic liquids: towards an extended Hofmeister series
Abstract
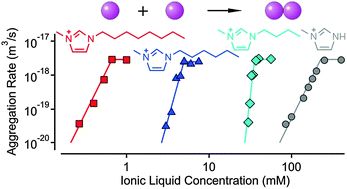
The effect of ionic liquid (IL) constituents and other monovalent salts on the stability of polystyrene latex particles was studied by electrophoresis and light scattering in dilute aqueous suspensions. The surface charge and the aggregation rate were both sensitive to the type of ion leading to different critical coagulation concentration (CCC) values. Systematic variation of the type of IL cation and anion allows us to place these ions within the Hofmeister series. We find that the dicyanoamide anion should be placed between iodide and thiocyanate, while all 1-alkyl-3-methylimidazolium cations can be positioned to the left of the tetramethylammonium and ammonium ions. The hydrophobicity of the 1-butyl-1-methylpyrrolidinium (BMPL+) ion is intermediate between 1-ethyl-3-methylimidazolium (EMIM+) and 1-butyl-3-methylimidazolium (BMIM+). With increasing alkyl chain length, the 1-alkyl-3-methylimidazolium cations adsorb on the latex particles very strongly, and 1-hexyl-3-methylimidazolium (HMIM+) and 1-octyl-3-methylimidazolium (OMIM+) lead to pronounced charge reversal and to an intermediate restabilization region.

 Please wait while we load your content...
Please wait while we load your content...