Conformational polymorphism of a twisted conjugated molecule: controllable torsional motion for crystal growth selectivity†
Abstract
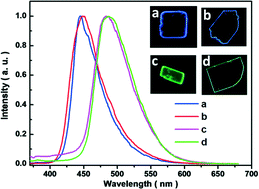
Controlling the emergence of the desired polymorphic form in crystal growth is of importance for both understanding the phenomenon of polymorphism and practical applications, but remains a great challenge as well. Herein, two polymorphs of cyano substituted distyrylbenzene (CN-DSB) are obtained as the crystals grow from chlorobenzene solution (Form I, blue fluorescence) and from its vapor (Form II, green fluorescence). Single-crystal X-ray analysis for the two crystal forms shows unambiguous evidence that the distinctive feature of molecules in both crystal forms is their difference in torsion angles of peripheral phenyl segments. A computational study addresses that the controllable growth of polymorphs can be rationalized on the basis of structural relationships with torsion potential under different external conditions. Our demonstration of single-crystal growth by tuning subtle external conditions illustrates a controllable process of obtaining conformational polymorphs.

 Please wait while we load your content...
Please wait while we load your content...