Highly selective Markovnikov hydroboration of alkyl-substituted terminal alkenes with a phosphine–copper(i) catalyst†
Abstract
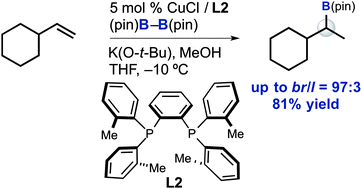
A new method has been developed for the Markovnikov hydroboration of alkyl-substituted terminal alkenes. Notably, the use of a bulky bisphosphine–copper(I) catalyst system resulted in high regioselectivity to afford secondary alkylboronates from the corresponding terminal alkenes (branch/linear = 92 : 8–97 : 3). This method also exhibited good functional group compatibility.

 Please wait while we load your content...
Please wait while we load your content...