Determination of reducing sugars in foodstuff applying multivariate second-order calibration†
Abstract
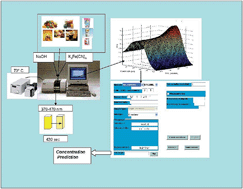
In the present report, a chemometrics-assisted second-order kinetic-spectrophotometric method has been developed for determining reducing sugars, glucose, fructose and lactose, in food samples, based on the reaction with hexacyanoferrate, HCF, at 70 °C in alkaline medium. A suitable experimental design helped us to establish the conditions (pH, temperature, and HCF concentration) for optimal sensitivity and selectivity among analytes. Second order data were recorded by measuring the absorbance of unreacted HCF in the spectral range of 370 to 470 nm for five minutes using a diode array. A calibration set of samples was prepared according to a central composite design containing the three sugars for training the algorithms. Validation samples containing only the analytes were prepared for checking the reliability of the algorithms. In this particular system, identical profiles for sample components are obtained in the spèctral dimension corresponding to unreacted HCF. Moreover, two kinds of interferents may be present: sample components active in the spectral region at which HCF absorbs as well as potential reducing interferents, causing linear dependence, since they provide identical profiles in spectral dimension to those of the analytes of interest. In the present work, MCR-ALS in the spectral augmentation mode was the only algorithm that could successfully resolve linear dependence. Satisfactory results were obtained by applying MCR-ALS in the spectral augmentation mode in order to achieve a second order advantage for the determination of fructose and glucose in validation samples, in test samples containing the two kinds of interferents and in real food samples, providing LODs of 4.0 and 5.0 mg L−1, respectively. However, bad results were obtained for lactose which may be due to its low sensitivity in the augmented dimension. Good results were also obtained by applying U-PLS/RBL and N-PLS/RBL for determining simultaneously the three sugars in validation samples and in test samples containing only active spectral interferents. Finally, lactose and also, glucose and fructose, were successfully quantified in real milk samples, with LODmin = 1.0 mg L−1, 1.0 mg L−1 and 0.1 mg L−1 and LODmax = 3.5, 3.8 and 4.4 mg L−1, respectively, using UPLS/RBL, and LODmin of 1.3, 1.1 and 0.1 mg L−1 and LODmax of 4.0, 4.3 and 4.9 mg L−1 for lactose, glucose and fructose, respectively, for NPLS/RBL. Results for real samples in all cases were statistically comparable to those obtained by applying a reference method based on HPLC (High Performance Liquid Chromatography).

 Please wait while we load your content...
Please wait while we load your content...