Solid oxide cells with zirconia/ceria Bi-Layer electrolytes fabricated by reduced temperature firing†
Abstract
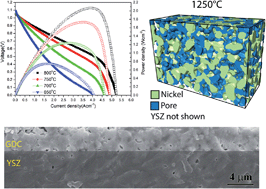
Anode-supported solid oxide cells (SOCs) with thin bi-layer Y0.16Zr0.92O2−δ (YSZ)/Gd0.1Ce0.9O1.95 (GDC) electrolytes were prepared by a reduced-temperature (1250 °C) co-firing process enabled by the addition of a Fe2O3 sintering aid. The Fe2O3 amounts in the layers affected the formation of voids at the GDC/YSZ interface; the case with 1 mol% Fe2O3 in the YSZ layer and 2 mol% Fe2O3 in the GDC layer yielded minimal interfacial voids, presumably because of optimized shrinkage matching between the electrolyte layers during co-firing. The best cells yield fuel cell power density at 0.7 V in air and humidified hydrogen of 1.74 W cm−2 (800 °C) and 1.0 W cm−2 (700 °C). Under electrolysis conditions, i.e., air and 50 vol% H2O–50 vol% H2, the best cell area specific resistance is 0.12 Ω cm2 at 800 °C and 0.27 Ω cm2 at 700 °C. This excellent cell performance was explained by a number of factors related to the reduced firing temperature: (1) low electrolyte resistance due to minimization of YSZ/GDC interdiffusion; (2) minimal zirconate phase formation between the YSZ and the La0.6Sr0.4Fe0.8Co0.2O3 (LSFC) cathode because of the dense GDC barrier layer; (3) high three phase boundary density in the Ni–YSZ anode functional layer; and (4) good pore connectivity in the Ni–YSZ support. Preliminary life testing under fuel cell and electrolysis operation shows promising cell stability.

 Please wait while we load your content...
Please wait while we load your content...