Secondary growth of hierarchical nanostructures composed only of Nb3O7F single-crystalline nanorods as a new photocatalyst for hydrogen production†
Abstract
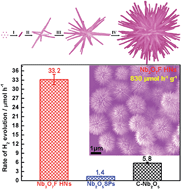
Development of efficient photocatalysts for hydrogen (H2) production from water splitting is highly sought for the current sustainable energy issue. This paper has demonstrated a one-pot hydrothermal approach for the preparation of hierarchical nanostructures (HNs) composed only of Nb3O7F single-crystalline nanorods by using H2NbF7 as a precursor and 2-propanol as a selective adsorption agent. In our protocol, modulating the hydrolysis rate of the coordination complex H2NbF7via simply controlling its concentration not only produces Nb3O7F single crystalline nanorods by the restrictive growth on [110] due to the selective adsorption of 2-propanol on the sidewalls, but also leads to secondary nucleation for the growth of new nanorods on the pre-formed ones, leading to the formation of Nb3O7F HNs. The resultant Nb3O7F HNs strongly respond to ultraviolet light and have a band gap of 3.22 eV as well as the conducting band minimum at −0.44 eV. They exhibit excellent photocatalytic H2 production activity with a good recycling stability when 0.5 wt% Pt is used as a cocatalyst. The H2 production rate reaches 830 µmol h−1 g−1, about 23.7 and 5.7 times higher than that of Nb2O5 sphere-shaped micro-particles and commercial Nb2O5 nanoparticles, respectively. Furthermore, they can also show notable photocatalytic H2 production activity even without any cocatalysts. This suggests that the resultant Nb3O7F HNs are promising as a new photocatalyst for H2 production.

 Please wait while we load your content...
Please wait while we load your content...