Adsorption of soft particles at fluid interfaces
Abstract
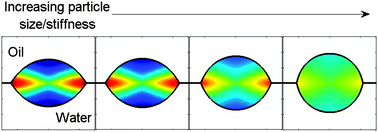
Soft particles can be better emulsifiers than hard particles because they stretch at fluid interfaces. This deformation can increase adsorption energies by orders of magnitude relative to rigid particles. The deformation of a particle at an interface is governed by a competition of bulk elasticity and surface tension. When particles are partially wet by the two liquids, deformation is localized within a material-dependent distance L from the contact line. At the contact line, the particle morphology is given by a balance of surface tensions. When the particle radius R ≪ L, the particle adopts a lenticular shape identical to that of an adsorbed fluid droplet. Particle deformations can be elastic or plastic, depending on the relative values of the Young modulus, E, and yield stress, σp. When surface tensions favour complete spreading of the particles at the interface, plastic deformation can lead to unusual fried-egg morphologies. When deformable particles have surface properties that are very similar to one liquid phase, adsorption can be extremely sensitive to small changes of their affinity for the other liquid phase. These findings have implications for the adsorption of microgel particles at fluid interfaces and the performance of stimuli-responsive Pickering emulsions.

 Please wait while we load your content...
Please wait while we load your content...