An exceptional functionalization of doped fullerene observed via theoretical studies on the interactions of sulfur-doped fullerenes with halogens and halides†
Abstract
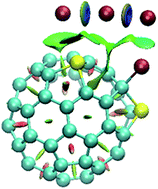
This work reports the study on the interactions of sulfur-doped fullerenes with halogens and halides (except iodine and iodide) to obtain a deep understanding of such interactions for employing them in possible applications such as sensors and surface adsorption. The ωB97XD DFT code has been utilized in this study to obtain adsorption energies in the gas phase and solvent. The energy outcomes showed good results for adsorption in both gas and solvent (using the PCM model). However, the gas phase interactions are more thermodynamically favorable. The formation of halide complexes releases more energy than the formation of halogen complexes and the strongest interaction belongs to the interactions of disulfur-doped fullerenes with halides. Donor–acceptor transitions are mostly affected by sulfur doping, which made the C–S bond an auxiliary tool for the absorption process. Density of state (DOS) plots demonstrate the enhanced modification of conductivity properties upon sulfur-doping on fullerene structures. Electron densities and their Laplacians at bond critical points (BCPs) of interaction sites, NCI calculations and further visualizations clearly prove the existence of these interactions. It is worth noting that, in some cases, some observations like the partial functionalization of fullerene was seen and these observations were proven via QTAIM, NCI and energy data. In these cases, the stable and thermodynamically favorable cation of the halogenated doped fullerene (along with X3− as the counter ion) could be produced from the interaction of double-doped fullerene with two molecular halogens.

 Please wait while we load your content...
Please wait while we load your content...