Monosubstitution at the 4-position of 2,7-carbazolylene expands the structural design and fundamental properties of D-π-A copolymers for organic photovoltaic cells†
Abstract
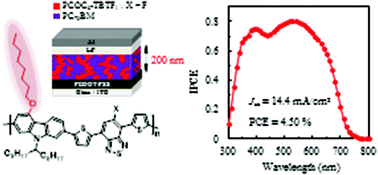
A 4-octyloxy-carbazole-2,7-diyl unit was newly developed, and was combined with dithienylbenzothiadiazole derivatives by using the Suzuki-coupling reaction to synthesize good processable donor-π-acceptor type copolymers. The monosubstitution at the 4-position of carbazolylene was not only effective to solubilize the narrow band gap copolymers in usual organic solvents but also could greatly improve their processability for device fabrication. All the polymers maintained the fundamental properties of carbazole–dithienylbenzothiadiazole copolymers such as good thermal stability, a narrow band gap of about 1.8 eV, a polymer interchain distance of 16–17 Å, a π-stacking distance of 3.5–4.1 Å and hole mobilities of 8.6 × 10−4–5.5 × 10−3 cm2 V−1 s−1. A bulk heterojunction organic photovoltaic device composed of the fluorinated copolymer and fullerene (PC70BM) with the active layer thickness of about 100 nm exhibited a power conversion efficiency (PCE) of 4.4% with a short-circuit current density (Jsc) of 11.6 mA cm−2, a fill factor of 0.56 and an open circuit voltage of 0.670 V. This polymer based device could sustain its photovoltaic performance even at a larger thickness of the active layer of about 200 nm, i.e. a PCE of 4.50% with a Jsc of 14.44 mA cm−2. The maximum incident photon to current conversion efficiency of this device was 80% at about 530 nm, which was the highest among other carbazole-copolymer-based devices.

 Please wait while we load your content...
Please wait while we load your content...