Well-defined polymethylene-based block co/terpolymers by combining anthracene/maleimide diels–alder reaction with polyhomologation†
Abstract
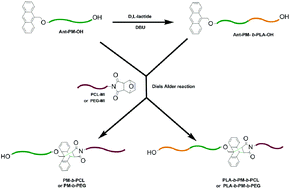
A novel strategy towards well-defined polymethylene-based co/terpolymers, by combining the anthracene/maleimide Diels–Alder reaction with polyhomologation, is presented. For the synthesis of diblock copolymers the following approach was applied: (a) synthesis of α-anthracene-ω-hydroxy-polymethylene by polyhomologation using tri(9-anthracene-methyl propyl ether)borane as the initiator, (b) synthesis of furan-protected-maleimide-terminated poly(ε-caprolactone) or polyethylene glycol and (c) the Diels–Alder reaction between the anthracene and maleimide-terminated polymers. In the case of triblock terpolymers the α-anthracene-ω-hydroxy-polymethylene was used as a macroinitiator for the ring-opening polymerization of D,L-lactide to afford an anthracene-terminated PM-b-PLA copolymer, followed by the Diels–Alder reaction with furan-protected maleimide-terminated poly (ε-caprolactone) or polyethylene glycol to give the triblock terpolymers. All the intermediates and final products were characterized by SEC, 1H NMR, UV-VIS spectroscopy and DSC.

 Please wait while we load your content...
Please wait while we load your content...