The leading role of cation–π interactions in polymer chemistry: the control of the helical sense in solution†
Abstract
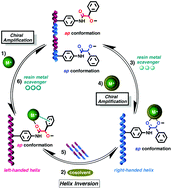
Cation–π interactions determine the helical sense adopted by a polyphenylacetylene bearing (R)-α-methoxy-α-phenylacetamide as a pendant group (poly-1). These interactions take place when small amounts of Li+, Na+ and Ag+ salts dissolved in donor cosolvents are added to the polymer dissolved in a non-donor solvent and the corresponding helical polymer–metal complexes (HPMCs) are formed. Extensive spectroscopic studies (i.e., 7Li, 23Na, 1H NMR and IR among others) indicate that the pendants act as dipodal receptors for metal cations, where cation–π interactions play a major role in controlling the M or P helicity of the polymer. Although all the cations tested generate cation–π binding, each metal presents its own particular and distinctive behaviour, based on polymer/cosolvent ratios and the type of cosolvent. Manipulation of these variables allows the selective disruption or activation of the cation–π interactions (i.e., a controlled switch on/off mechanism) and the selection of the helical sense on an à la carte basis. The 3/1 helical structure of poly-1 can cause a novel “triple cascade effect” of cooperative cation–(π)n–π interactions, which further induce the high chiral amplification response found in these HPMCs. These non-covalent interactions are not found when divalent cations are used instead.

 Please wait while we load your content...
Please wait while we load your content...