Amphiphilic glycosylated block copolypeptides as macromolecular surfactants in the emulsion polymerization of styrene†
Abstract
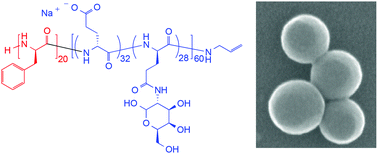
Diblock copolymers consisting of poly(L-phenyl alanine) and poly(benzyl-L-glutamate) or poly(CBZ-L-lysine), respectively, were synthesized via sequential NCA polymerization. After deprotection, subsequent partial glycosylation of the glutamic acid and lysine units with galactosamine hydrochloride or lactobionic acid yielded amphiphilic block copolypeptides. Moreover, a triblock copolymer poly(L-phenyl alanine-b-L-benzyl glutamate-b-propargylglycine) was obtained and glycosylated by ‘click’ chemistry. Glycosylated block copolypeptides showed improved water solubility and circular dichroism (CD) confirmed the pH dependence of the helix-coil transition. The block copolypeptides were found to be efficient stabilizers in the emulsion polymerization of styrene offering a facile method for the synthesis of polystyrene nanoparticles in the range of 100–140 nm depending on the block copolymer composition and emulsion concentration. This establishes an example of functional polymer additives fully based on renewable building blocks in nanomaterial synthesis.

 Please wait while we load your content...
Please wait while we load your content...