Hybridization of zirconia, zinc and iron supported on HY zeolite as a solar-based catalyst for the rapid decolorization of various dyes
Abstract
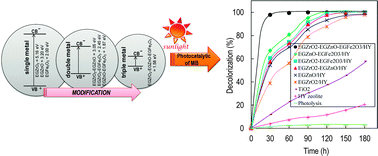
Hybridization of an electrogenerated zirconia/zinc/iron-supported on HY zeolite nanocomposite catalyst (EGZrO2/EGZnO/EGFe2O3/HY) was performed by a facile one-pot electrochemical method. In this study, nanoparticles (<35 nm in size) of electrogenerated zirconia oxide (EGZrO2), electrogenerated zinc oxide (EGZnO), and electrogenerated iron oxide (EGFe2O3) were obtained and distributed on the surface of a HY support. The photoactivity of the catalyst was examined for the photodecolorization of methylene blue (MB) under varying pH values, catalyst dosages, and initial concentrations of MB. 0.38 g L−1 of 1 wt% EGZrO2/1 wt% EGZnO/1 wt% EGFe2O3/HY was found to be the optimum dosage for 10 mg L−1 MB, which gave the complete decolorization of MB after 1 h of contact time at pH 9 under sunlight irradiation. This result confirmed the high photoactivity of the catalyst compared to other single or double nanometal oxide-supported HY catalysts studied. Nearly complete mineralization of MB was observed, and the catalyst was still stable after six cycling runs with a negligible leaching effect. Significantly, a high decolorization percentage (>80%) of other dyes, such as malachite green (MG), congo red (CR), disperse violet (DV), and disperse blue (DB), were also obtained using this promising electrogenerated nanocomposite catalyst.

 Please wait while we load your content...
Please wait while we load your content...