Influence of calcination on performance of Bi–Ni–O/gamma-alumina catalyst for n-butane oxidative dehydrogenation to butadiene†
Abstract
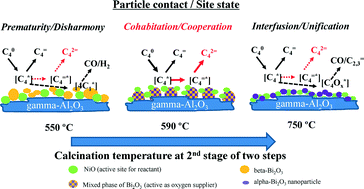
Influence of calcination on the binary-metal oxide 30 wt% Bi-20 wt% Ni–O/gamma-Al2O3 catalyst was studied for n-butane oxidative dehydrogenation to 1,3-butadiene. The importance of the calcination was confirmed with the facts that (1) the two step calcination of more than 2 h at the 2nd step resulted in higher activity and selectivity to butadiene and (2) the second step calcination temperatures showed clear effects on the activity and the selectivity. The activity showed a volcano shaped trend, with the top at 590 °C and at a lower reaction temperature, 400 °C, and a downward slope at 450 °C. As for the reaction selectivity, either the total dehydrogenation or the specific butadiene selectivity showed a volcano shape with the calcination temperature. The top along the calcination temperature existed at 590 °C in both cases of 400 °C and 450 °C reaction temperatures. Conversely, the selectivity of oxygenate formation (partially followed by cracking) or partial oxidation to CO/H2 showed a valley shape with the bottom at 590 °C or 650 °C along the calcination temperature, respectively. The butadiene selectivity was more strongly influenced competitively by both the oxygenate formation and the partial oxidation. From the catalyst characterization, it was observed that the redox and acid/base system (active and selective to the reaction) of the combined oxides with porous structure are formed as a cohabitation consisting of ‘hierarchical nanoparticles’ (NiO, alpha-/beta-Bi2O3 and gamma-Al2O3), resulting from catalyst calcination in preferable conditions.

 Please wait while we load your content...
Please wait while we load your content...