Transient low-barrier hydrogen bond in the photoactive state of green fluorescent protein
Abstract
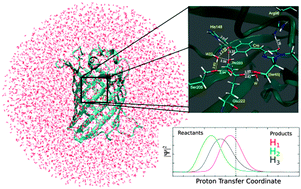
In this paper, we have analyzed the feasibility of spontaneous proton transfer in GFP at the Franck–Condon region directly after photoexcitation. Computation of a sizeable portion of the potential energy surface at the Franck–Condon region of the A structure shows the process of proton transfer to be unfavorable by 3 kcal mol−1 in S1 if no further structural relaxation is permitted. The ground vibrational state is found to lie above the potential energy barrier of the proton transfer in both S0 and S1. Expectation values of the geometry reveal that the proton shared between the chromophore and W22, and the proton shared between Ser205 and Glu222 are very close to the center of the respective hydrogen bonds, giving support to the claim that the first transient intermediate detected after photoexcitation (I0*) has characteristics similar to those of a low-barrier hydrogen bond [Di Donato et al., Phys. Chem. Chem. Phys., 2012, 13, 16295]. A quantum dynamical calculation of the evolution in the excited state shows an even larger probability of finding those two protons close to the center compared to in the ground state, but no formation of the proton-transferred product is observed. A QM/MM photoactive state geometry optimization, initiated using a configuration obtained by taking the A minimum and moving the protons to the product side, yields a minimum energy structure with the protons transferred and in which the His148 residue is substantially closer to the now anionic chromophore. These results indicate that: (1) proton transfer is not possible if structural relaxation of the surroundings of the chromophore is prevented; (2) protons H1 and H3 especially are found very close to the point halfway between the donor and acceptor after photoexcitation when the zero-point energy is considered; (3) a geometrical parameter exists (the His148–Cro distance) under which the structure with the protons transferred is not a minimum, and that, if included, should lead to the fluorescing I* structure. The existence of an oscillating stationary state between the reactants and products of the triple proton transfer reaction can explain the dual emission reported for the I0* intermediate of wtGFP.
- This article is part of the themed collection: Measurement and prediction of quantum coherence effects in biological processes


 Please wait while we load your content...
Please wait while we load your content...