Cationic surfactant-assisted hydrothermal synthesis: an effective way to tune the crystalline phase and morphology of SAPO molecular sieves†
Abstract
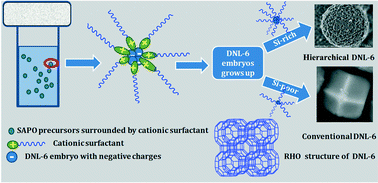
The present work investigates the effect of cationic surfactants on tuning the crystalline phase and morphology of SAPO molecular sieves. First, we explore the cationic surfactant-assisted hydrothermal synthesis of DNL-6, which is a newly developed SAPO molecular sieve with RHO structure and can be prepared only in a narrow silica concentration. Comparative experiments reveal that the cationic surfactant with a longer alkane chain can help to obtain pure DNL-6 with an adjustable Si content, and the product with a higher Si content presents a spherical morphology composed of DNL-6 nanocrystals. It is believed that the strong adsorption of the cationic surfactant molecules on the SAPO species protects the DNL-6 from dissolving and inhibits the crystallization of SAPO-34. It is thus concluded that the cationic surfactant favors the formation of SAPO crystals having a relatively high structural symmetry although the surfactants are not retained in the product. Moreover, the crystal morphology may be modified due to the abundant interactions of the cationic surfactant with the anionic SAPO framework. Based on the above understanding, flower-like SAPO-35 and Si-rich SAPO-16 were further synthesized by designed cationic surfactant-assisted syntheses. These confirm that the cationic surfactant-assisted hydrothermal synthesis has huge potential in the control of the SAPO crystalline phase and crystal morphology. More importantly, the obtained hierarchical porous DNL-6 exhibits high catalytic activity and selectivity for the cycloaddition of CO2 with epichlorohydrin without any solvent and co-catalyst.

 Please wait while we load your content...
Please wait while we load your content...