A straightforward implementation of in situ solution electrochemical 13C NMR spectroscopy for studying reactions on commercial electrocatalysts: ethanol oxidation
Abstract
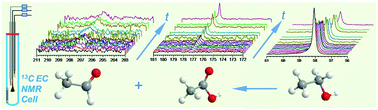
Identifying and quantifying electrocatalytic-reaction-generated solution species, be they reaction intermediates or products, are highly desirable in terms of understanding the associated reaction mechanisms. We report herein a straightforward implementation of in situ solution electrochemical 13C NMR spectroscopy for the first time that enables in situ studies of reactions on commercial fuel-cell electrocatalysts (Pt and PtRu blacks). Using ethanol oxidation reaction (EOR) as a working example, we discovered that (1) the complete oxidation of ethanol to CO2 only took place dominantly at the very beginning of a potentiostatic chronoamperometric (CA) measurement and (2) the PtRu had a much higher activity in catalysing oxygen insertion reaction that leads to acetic acid.

 Please wait while we load your content...
Please wait while we load your content...