A Lewis acid/metal amide hybrid as an efficient catalyst for carbon–carbon bond formation†
Abstract
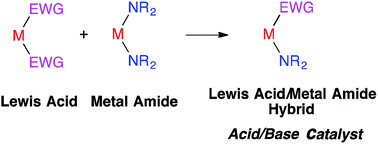
While Lewis acids and metal amides are among the most frequently used metal species, they are believed to be incompatible when combined. Here we describe a Lewis acid/metal amide hybrid, which contains electron-withdrawing groups and basic and bulky nitrogen functional groups in the same metal complex, as a novel catalyst. We have synthesized In(N(SiMe3)2)2Cl (In(HMDS)2Cl) and In(HMDS)2OTf as Lewis acid/metal amide hybrids, which showed excellent catalytic activity for the reaction of nitrones with terminal alkynes to give synthetically useful propargyl hydroxylamines. It is noted that neither the Lewis acids (InCl3, In(OTf)3) nor the metal amides (In(HMDS)3) have activity; only the hybrids worked well, and the catalytic activity of the hybrids was shown to be much higher than that of previously reported catalysts for this reaction. The concept of a Lewis acid/metal amide hybrid as a catalyst may be expanded to broad acid/base catalysis.

 Please wait while we load your content...
Please wait while we load your content...