Facile RAFT synthesis of side-chain amino acids containing pH-responsive hyperbranched and star architectures†
Abstract
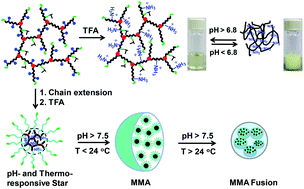
This work reports the design and synthesis of amino acid-based hyperbranched polymers via the combination of self-condensing vinyl polymerization (SCVP) and reversible addition–fragmentation chain transfer (RAFT) polymerization from tert-butyl carbamate (Boc)-L-valine acryloyloxyethyl ester (Boc-Val-HEA) and S-(4-vinyl)benzyl S′-butyltrithiocarbonate (VBBT) with variable degrees of branching (DB), molecular weights (Mn), and chain end functionalities. Copolymerization kinetics reveal that the molecular weight increases and the DB decreases linearly with time as the branch length increases with the conversion of the Boc-Val-HEA monomer. These hyperbranched polymers, P(Boc-Val-HEA-co-VBBT), with tuneable Mn and DB have been further employed via successive RAFT polymerizations for the synthesis of star polymers with variable arm numbers and lengths. The removal of Boc groups from the polymers results in water soluble pH-responsive cationic hyperbranched architectures with tuneable pH responsiveness, differing from 6.8–7.5 due to the incorporation of various degrees of hydrophobic chain end functionalities with the variation of monomer feed compositions. Dynamic light scattering (DLS), atomic force microscopy (AFM) and scanning electron microscopy (SEM) reveal the interesting self-assembly of the Boc-deprotected star polymers in aqueous media with amino acid-based cores and water soluble thermoresponsive arms. Below the hydrophilic to hydrophobic transition pH and temperature, star polymers remain as unimers in aqueous solution. However, above the transition pH (and below the transition temperature), they form multi-micellar aggregates, which further fuse together to form larger aggregates above the transition temperature.

 Please wait while we load your content...
Please wait while we load your content...