NH-type of chiral Ni(ii) complexes of glycine Schiff base: design, structural evaluation, reactivity and synthetic applications†
Abstract
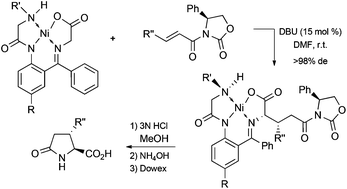
The work being reported here deals with the design of a new type of “N–H” Ni(II) complexes of glycine Schiff bases and study general aspects of their reactivity. It was confirmed that the presence of NH function in these Ni(II) complexes does not interfere with the homologation of the glycine residue, rendering these derivatives of high synthetic value for the general synthesis of α-amino acids. In particular, the practical application of these NH-type complexes was demonstrated by asymmetric synthesis of various β-substituted pyroglutamic acids via Michael addition reactions with chiral Michael acceptors.

 Please wait while we load your content...
Please wait while we load your content...