Trifluoromethanesulfonyloxy-group-directed regioselective (3 + 2) cycloadditions of benzynes for the synthesis of functionalized benzo-fused heterocycles†
Abstract
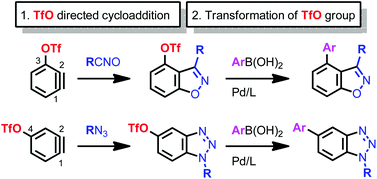
Highly regioselective (3 + 2) cycloadditions of (trifluoromethanesulfonyloxy)benzynes [(triflyloxy)benzynes] with 1,3-dipoles followed by cross-coupling reactions provided multisubstituted benzo-fused heterocycles. The triflyloxy group at the 3-position of benzynes, and even that at the remote 4-position, greatly affected the regiocontrol of the cycloaddition. These groups also served to install other substituents at their ipso-positions.

 Please wait while we load your content...
Please wait while we load your content...