A computational study of the activation of allenoates by Lewis bases and the reactivity of intermediate adducts†
Abstract
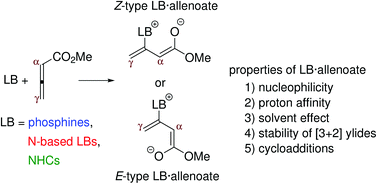
Several chemical properties of Lewis base-allenoate adducts (LB·allenoate), such as solvent effect, basicity, nucleophilicity and cycloaddition, are studied to provide a detailed foundation for the analysis of LB-catalyzed reactions of allenoates. The zwitterionic LB·allenoates formed between methyl allenoate and Lewis bases, such as N-heterocyclic carbenes (NHCs), phosphines, amines and aza-heterocycles, are studied at the M06-2X/6-31+G* level. The addition of the LBs to the allenoate can yield Z- or E-type adducts. The formation of the Z-type adducts is more favorable in the gas phase due to electrostatic interactions. The yield of the E-type adducts increases with the permittivity of the solvent. The lowest barriers for the addition and the most stable adducts are observed with NHCs as catalysts. It is also shown that the α-carbon atom of the allenic moiety in LB·allenoate is more nucleophilic than the γ-carbon atom. Aza-arenes, phosphines and NHCs stabilize the [3 + 2]-ylides formed by the cycloaddition of LB·allenoate to ethylene; therefore, these LBs thermodynamically support the [3 + 2] cycloadditions. The detailed analysis of [3 + 2]-, [2 + 4]-, [2 + 2]- and [2 + 2 + 2]-cycloadditions with enones/ketones shows that the amine-catalyzed reactions follow the kinetically preferred path, and that the exergonic formation of the P-ylide favors the [3 + 2] cycloaddition in the phosphine-catalyzed reaction. The thermodynamically preferred pathway is followed with NHCs whereas the high stability of NHC·allenoate adducts reduces the overall catalytic efficiency of NHCs.

 Please wait while we load your content...
Please wait while we load your content...