Synthesis, structural characterization and reactivity of heteroazuliporphyrins†
Abstract
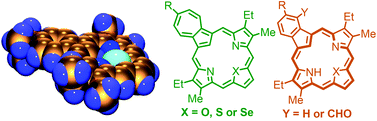
A series of hetero-azuliporphyrins have been prepared by the “3 + 1” variant on the MacDonald condensation. Azulitripyrranes with tert-butyl and phenyl substituents reacted with thiophene or selenophene dialdehydes in the presence of TFA to give, following an oxidation step, thia- and selena-azuliporphyrins in 45–55% yield. Two of these compounds gave crystals suitable for X-ray crystallographic analysis and the data were consistent with the presence of a 17-atom delocalization pathway. The hetero-azuliporphyrins have significant diatropic character that is enhanced by the presence of an electron-donating tert-butyl substituent. The aromatic character is further increased in polar solvents such as DMSO, which are believed to stabilize dipolar resonance contributors with 18π electron delocalization pathways. Protonation also greatly increases the diatropic characteristics of these macrocycles. The porphyrinoids underwent an oxidative ring contraction with t-BuOOH–KOH to give moderate yields of benzoheterocarbaporphyrins. Reaction of azulitripyrranes with 2,5-furandicarbaldehyde afforded oxa-azuliporphyrins, a class of carbaporphyrinoids that had previously been inaccessible. These “missing links” in the study of heteroazuliporphyrins were isolated as the dihydrochloride salts. Protonated oxa-azuliporphyrins are stable aromatic compounds, but the free base forms underwent rapid decomposition in solution.

 Please wait while we load your content...
Please wait while we load your content...